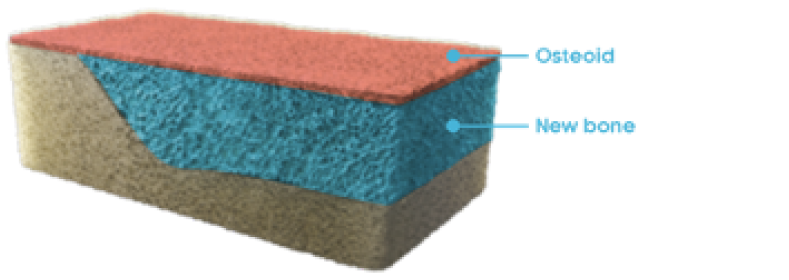
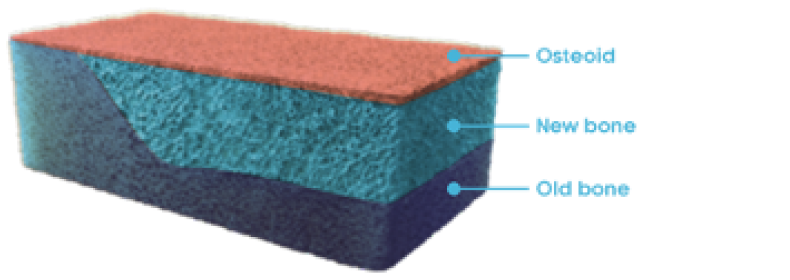
Osteomalacia and bone histomorphometry overview
Efficacy in adults
Actor portrayal

Clinical studies showed that CRYSVITA1:
- Increased and maintained serum phosphorus levels1
- Healed fractures and osteomalacia1
Study design
Study 41,2
CRYSVITA was studied in a randomized, double-blind, placebo-controlled phase 3 study in 134 adult patients with XLH. Patients were randomized to receive CRYSVITA 1 mg/kg every 4 weeks (n=68) or placebo (n=66) for 24 weeks, followed by a 24-week open-label period during which all patients received CRYSVITA.
- Proportion of subjects achieving mean serum phosphorus levels above the lower limit of normal at the midpoint of dosing interval, averaged across dose cycles from baseline to week 24
- Change from baseline to week 24 in patient-reported joint stiffness, as assessed by the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC)
- Resolution of pre-existing active pseudofractures and/or fractures at postbaseline visits, as defined by skeletal survey
Study 51,3
CRYSVITA was studied in a 48-week, open-label, single-arm study in 14 adult patients with XLH. Patients received CRYSVITA 1 mg/kg every 4 weeks.
- Percent change from baseline to week 48 in osteoid volume to bone volume as determined by iliac crest biopsies
- Percent change from baseline in additional histomorphometric parameters, including:
- - Osteoid thickness
- - Mineralization lag time
In both studies of adult patients with XLH, oral phosphate and active vitamin D analogs were not allowed.1-3
Disease burden at baseline1-3
CHANGE IN SERUM PHOSPHORUS LEVELS*
More patients on CRYSVITA increased and maintained serum phosphorus levels over 24 weeks1,2
CRYSVITA
(95% CI: 85.8, 97.7)
Placebo
(95% CI: 3.3, 16.5)
*The primary efficacy endpoint was the proportion of patients achieving a mean serum phosphorus concentration above the LLN of 2.5 mg/dL (0.81 mmol/L) averaged across the midpoints of dosing intervals.2
†P-value is from Cochran-Mantel-Haenszel (CMH) testing for association between achieving the primary endpoint and treatment group, adjusting for randomization stratification.1
Through week 24, 94% (64 out of 68) of patients achieved mean serum phosphorus levels above the lower limit of normal (LLN)‡ of 2.5 mg/dL (0.81 mmol/L), compared with 8% (5 out of 66) of patients receiving placebo. Serum phosphorus was maintained with continued CRYSVITA treatment through week 48.1,2
‡LLN is 2.5 mg/dL. The range of normal levels of serum phosphorus is 2.5 mg/dL to 4.5 mg/dL. Note that the range of normal levels of phosphorus differ based on age and sex.1,4,5
CI=confidence interval.
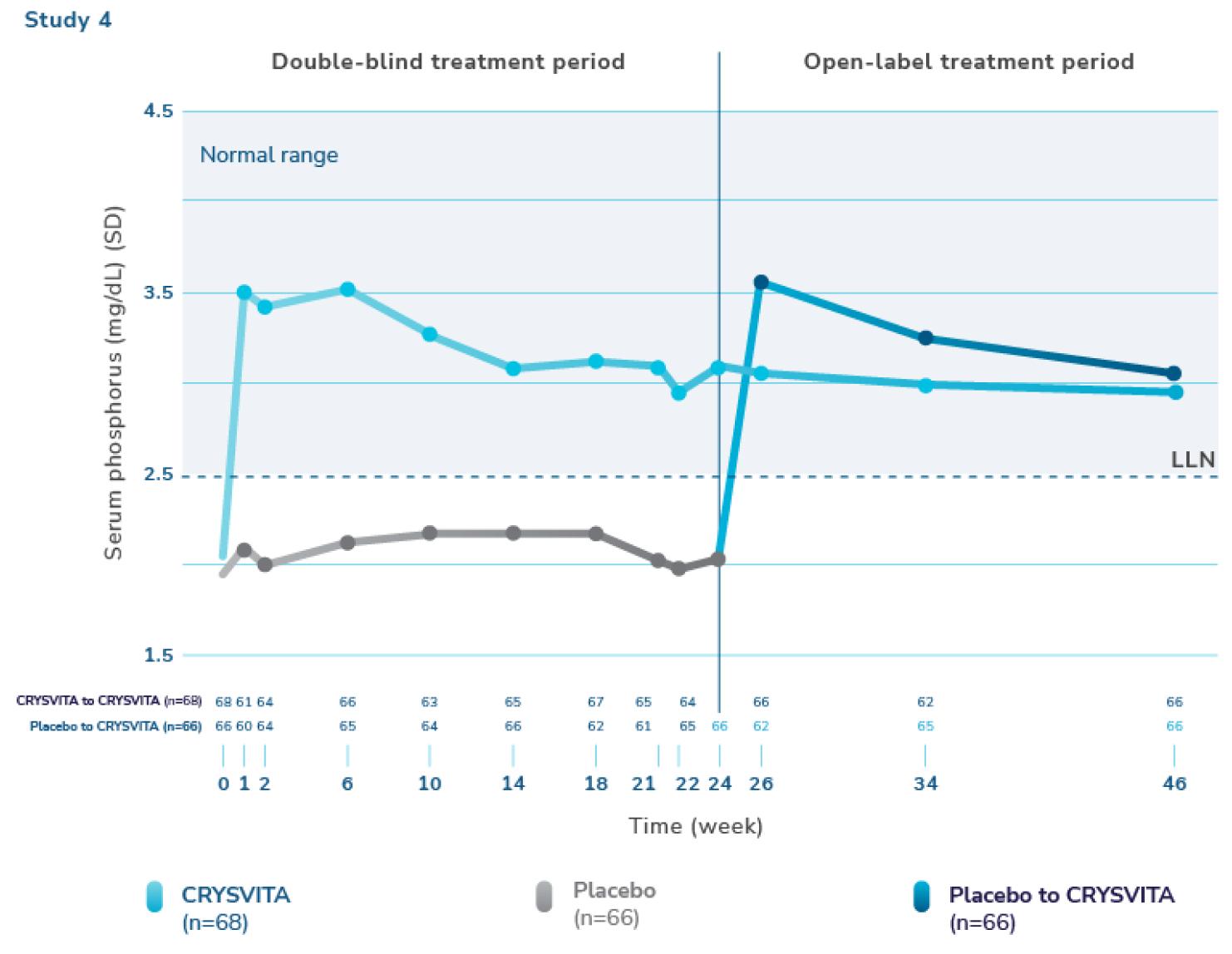
CRYSVITA increased serum phosphorus levels within the normal range1,2
§ Serum phosphorus level (mg/dL) (mean ±SD). The dotted line represents the lower limit of normal (LLN, 2.5 mg/dL). Normal levels of serum phosphorus range from 2.5 to 4.5 mg/dL. Note that the normal levels of serum phosphorus vary by age and sex. At baseline, mean (SD) serum phosphorus levels were 2.0 (0.30) mg/dL and 1.9 (0.32) mg/dL for the CRYSVITA and placebo groups, respectively. Mean serum phosphorus levels across midpoints of dose intervals (2 weeks post-dose) were 3.2 (0.53) mg/dL and 2.1 (0.30) mg/dL for the CRYSVITA and placebo groups, respectively.1,4,5
SD=standard deviation.
In Study 4, there was a significant increase from baseline through week 24 in mean serum phosphorus levels with CRYSVITA compared with placebo. CRYSVITA-treated patients maintained mean serum phosphorus levels within the normal range through week 48.1
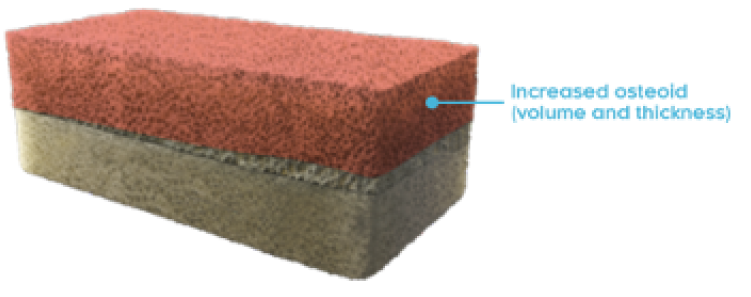
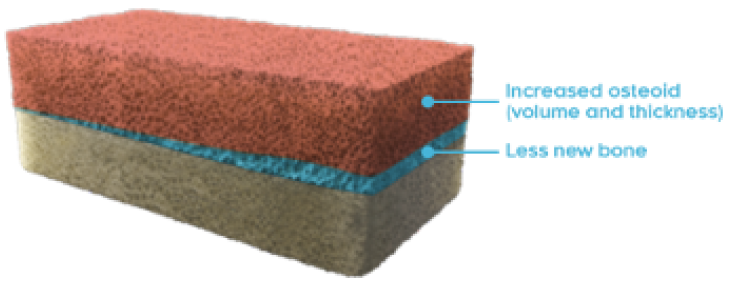
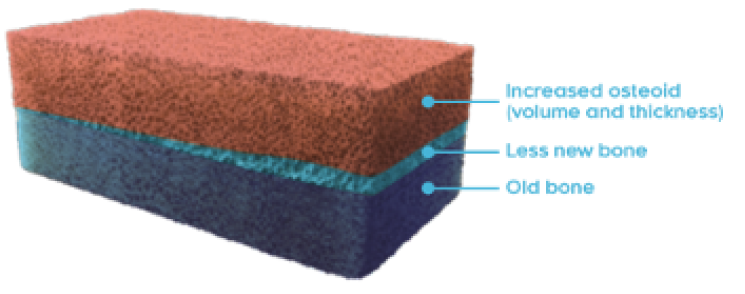
OSTEOMALACIA HEALING
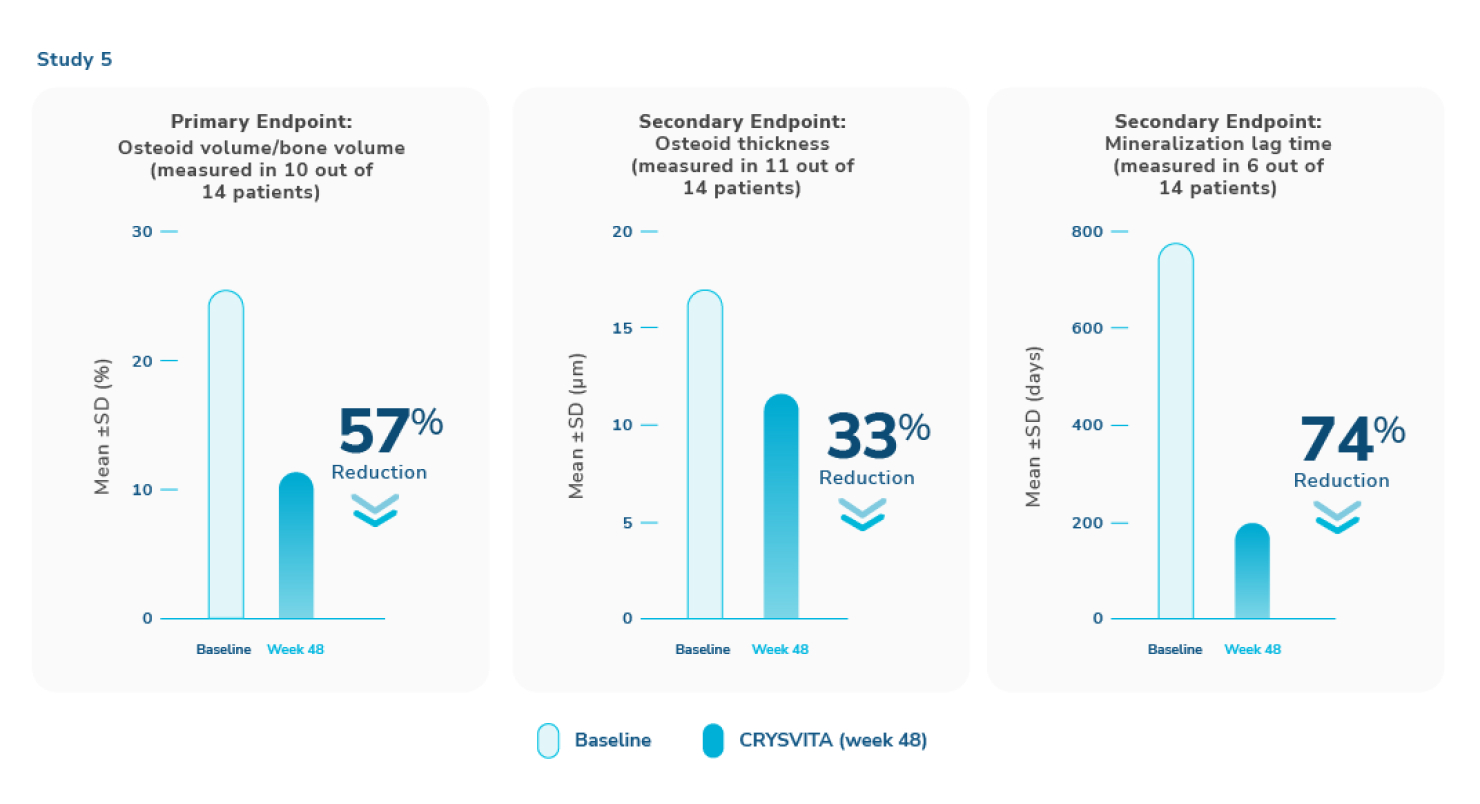
CRYSVITA improved the healing of osteomalacia1,3
‖ Normal osteoid volume to bone volume was defined as 0.30% to 3.10%; osteoid thickness as 5.5 to 12 μm; and mineralization lag time as 15 to 50 days.6
SD=standard deviation.
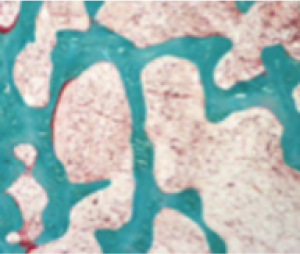
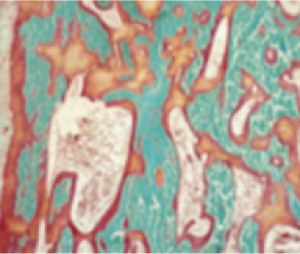
In Study 5, histological and histomorphometric assessments of iliac bone crest (biopsies) were examined for signs of osteomalacia healing. CRYSVITA improved healing at week 48, with significant reductions in osteoid volume to bone volume (OV/BV), osteoid thickness (O.Th), and mineralization lag time (MLt).1,3
In 10 patients, the mean (SD) score decreased in OV/BV from 26% (12.4) at baseline to 11% (6.5), a 57% reduction. O.Th declined in 11 patients from a mean (SD) of 17 (4.1) μm to 12 (3.1) μm, a 33% reduction. In 6 patients, MLt was reduced from a mean (SD) of 594 (675) days to 156 (77) days, a 74% decrease.1,3
FRACTURE HEALING
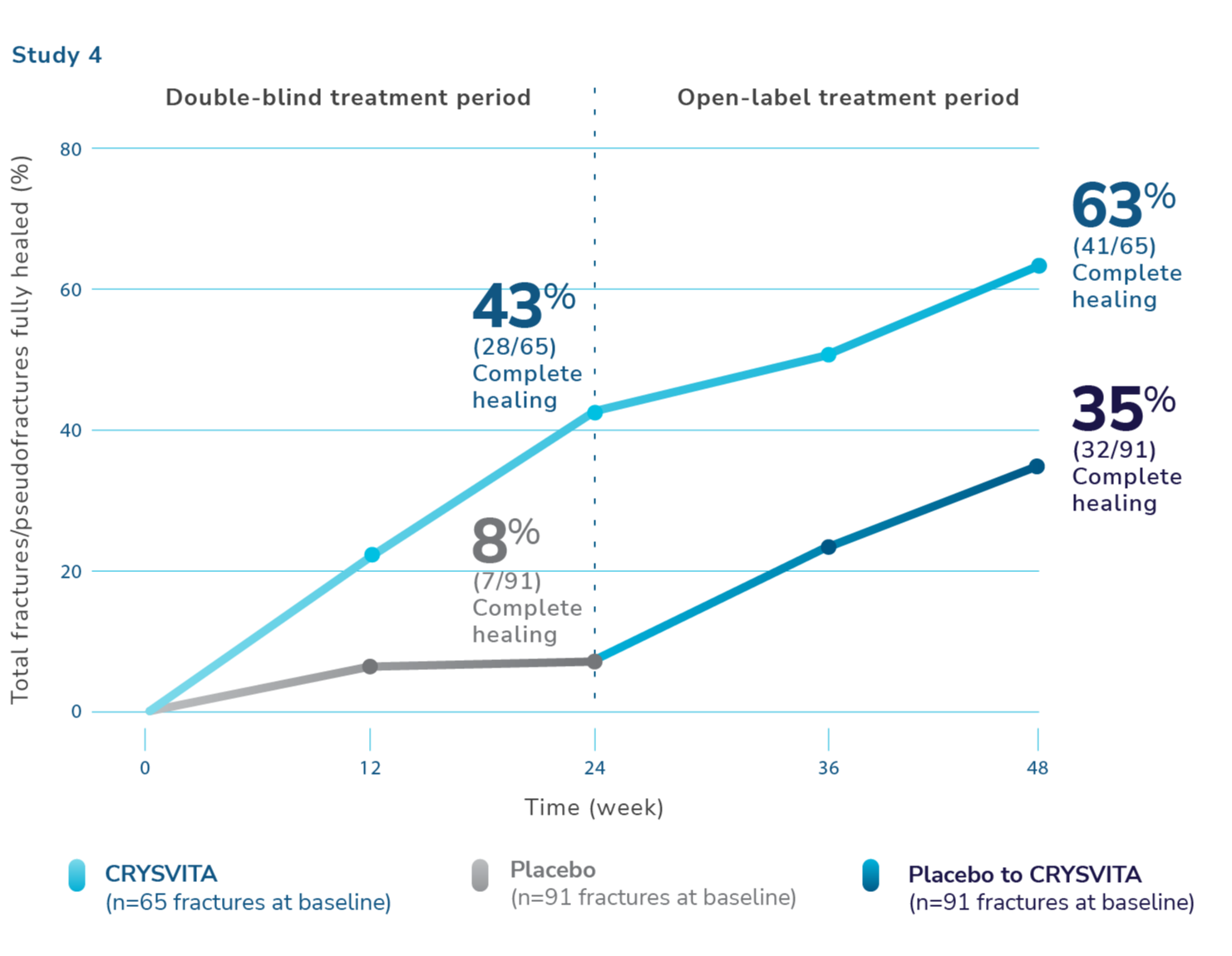
CRYSVITA helped heal osteomalacia-related fractures/pseudofractures1,2,7
¶Total fractures were both active fractures and pseudofractures. At baseline, the total number of active fractures/pseudofractures were 65 with CRYSVITA and 91 with placebo. Total fractures are osteomalacia-related fractures that were defined as atraumatic lucencies extending across both bone cortices, and pseudofractures that were defined as atraumatic lucencies extending across 1 cortex. These fractures were predominantly located in femurs, tibia/fibula, and metatarsals of the feet. Healing is defined as complete or partial healing.1,7
At week 24, 43% of baseline fractures/pseudofractures were healed in the CRYSVITA arm. During the open-label treatment period, patients who continued receiving CRYSVITA showed a higher rate of fracture healing with 63% of fractures/pseudofractures healed at week 48.1,2,7
At week 24, 68 patients receiving CRYSVITA had a total of 6 new fractures or pseudofractures compared with 8 new abnormalities in 66 patients receiving placebo.1
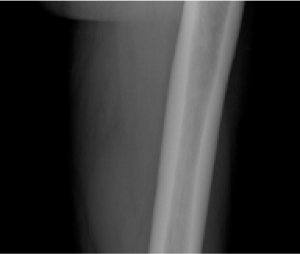
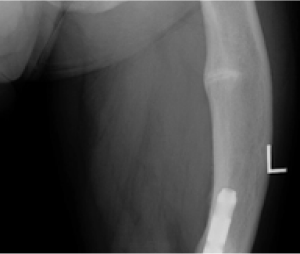
Radiographic examples of demonstrated osteomalacia-related pseudofracture healing2
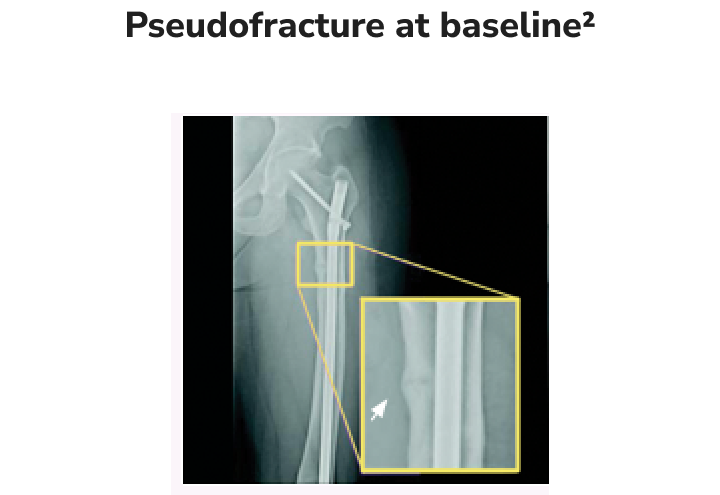
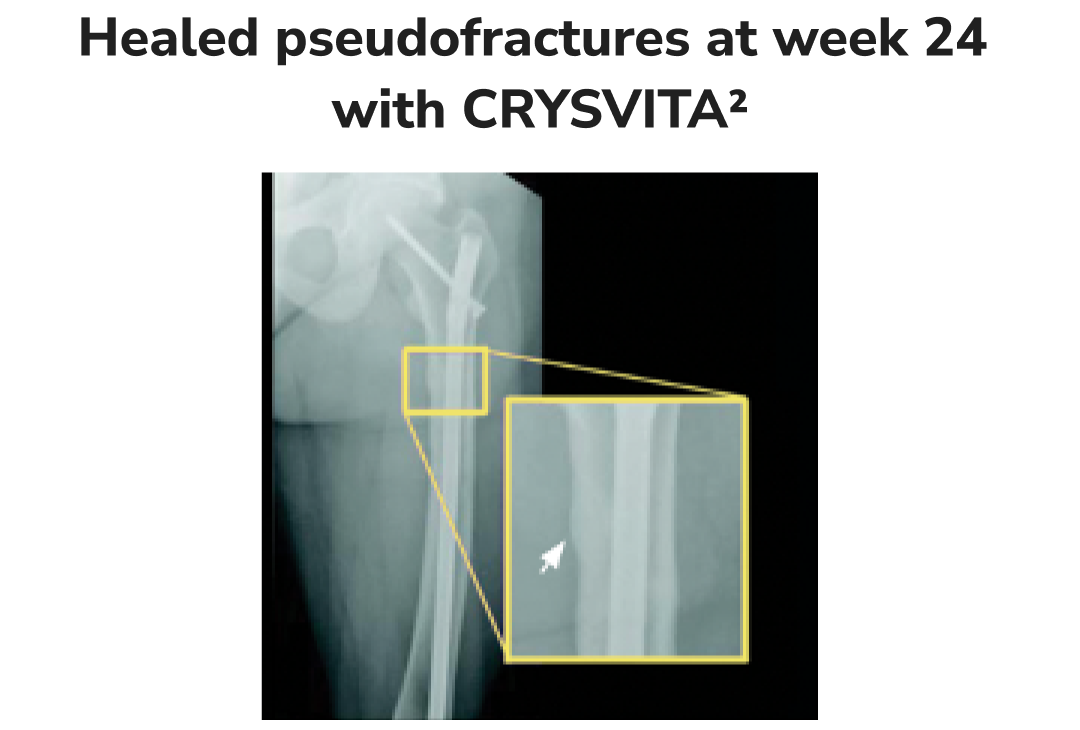
Individual results may vary.
PATIENT-REPORTED OUTCOMES
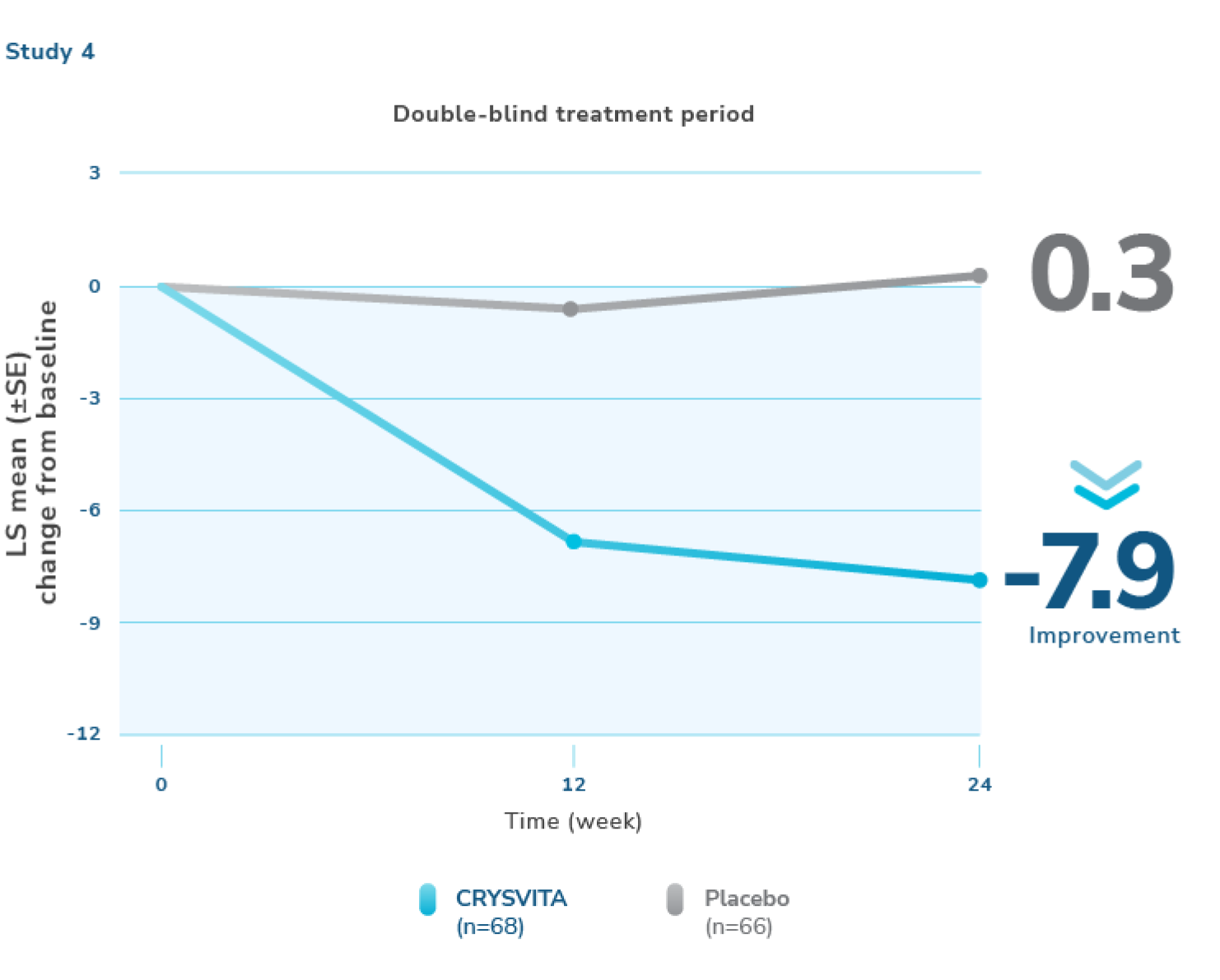
CRYSVITA was shown to improve XLH-related joint stiffness; no significant differences were noted in pain and physical function1,2
Study 4 evaluated patient-reported XLH-related symptoms (pain, joint stiffness, and physical function). CRYSVITA was shown to improve patient-reported joint stiffness compared with placebo; no significant differences were noted in patient-reported pain intensity or physical function score at 24 weeks. Patient-reported joint stiffness was assessed using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC).1,2
- The CRYSVITA arm showed a mean improvement from baseline (-7.9) compared with the placebo arm (+0.3) in the stiffness severity score (range 0 to 100; lower scores reflect less severe symptoms)
Mean (SD) baseline stiffness scores were 61.4 (20.77) and 64.7 (20.25) in the placebo and CRYSVITA groups, respectively.7
#The estimates of LS means at week 24 are from the generalized estimating equation (GEE) model.2
LS=least squares; SE=standard error; WOMAC=Western Ontario and McMaster Universities Osteoarthritis Index.

Ready to start your patients on CRYSVITA?
Take the first step by filling out the enrollment form.


Stay connected
Set up time with a representative to talk more about CRYSVITA,
or sign up for more information on CRYSVITA for the treatment of XLH.