



Tumor-induced osteomalacia, or TIO, is an extremely rare, difficult to diagnose disease—to date, there have only been about 1000 cases reported. The mean age of diagnosis is 45 years with a wide age range, including cases reported in children.2-4
TIO is an acquired form of hypophosphatemia caused by slow growing, benign, phosphaturic mesenchymal tumors that can be located anywhere in the body in soft tissues or bones. These tumors secrete excess fibroblast growth factor 23 (FGF23), which results in hypophosphatemia and osteomalacia.3,4
In normal homeostasis3,4:




Normal FGF23 activity

FGF23 plays an important role in phosphorus homeostasis via renal excretion


Intestinal phosphorus absorption also regulates phosphorus homeostasis

Normal bone mineralization
In TIO3-5:



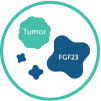

Tumors express increased production of FGF23
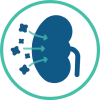

Excess FGF23 results in reduced renal phosphorus reabsorption or "phosphate wasting"


Reduced renal production of active vitamin D results in decreased intestinal phosphorus absorption

Resulting in chronic hypophosphatemia leading to osteomalacia
Tumor removal can be curative, but may not be possible under certain circumstances.2

Take the first step by filling out the enrollment form.

CRYSVITA® (burosumab-twza) is a fibroblast growth factor 23 (FGF23) blocking antibody indicated for the treatment of FGF23-related hypophosphatemia in tumor-induced osteomalacia (TIO) associated with phosphaturic mesenchymal tumors that cannot be curatively resected or localized in adult and pediatric patients 2 years of age and older.
CRYSVITA® (burosumab-twza) is a fibroblast growth factor 23 (FGF23) blocking antibody indicated for the treatment of FGF23-related hypophosphatemia in tumor-induced osteomalacia (TIO) associated with phosphaturic mesenchymal tumors that cannot be curatively resected or localized in adult and pediatric patients 2 years of age and older.
CONTRAINDICATIONS
CRYSVITA is contraindicated:
WARNINGS AND PRECAUTIONS
Hypersensitivity
Hyperphosphatemia and Risk of Nephrocalcinosis
Injection Site Reactions
ADVERSE REACTIONS
Adult Patients
USE IN SPECIFIC POPULATIONS
PATIENT COUNSELING INFORMATION
You may report side effects to the FDA at (800) FDA-1088 or www.fda.gov/medwatch. You may also report side effects to Kyowa Kirin, Inc. at 1-844-768-3544.
For important risk and use information, please see the full Prescribing Information for CRYSVITA.
CRYSVITA® (burosumab-twza) is a fibroblast growth factor 23 (FGF23) blocking antibody indicated for the treatment of FGF23-related hypophosphatemia in tumor-induced osteomalacia (TIO) associated with phosphaturic mesenchymal tumors that cannot be curatively resected or localized in adult and pediatric patients 2 years of age and older.
CONTRAINDICATIONS
CRYSVITA is contraindicated:
WARNINGS AND PRECAUTIONS
Hypersensitivity
Hyperphosphatemia and Risk of Nephrocalcinosis
Injection Site Reactions
ADVERSE REACTIONS
Adult Patients
USE IN SPECIFIC POPULATIONS
PATIENT COUNSELING INFORMATION
You may report side effects to the FDA at (800) FDA-1088 or www.fda.gov/medwatch. You may also report side effects to Kyowa Kirin, Inc. at 1-844-768-3544.
For important risk and use information, please see the full Prescribing Information for CRYSVITA.
References:
1. Farrow EG, White KE. Tumor-induced osteomalacia. Expert Rev Endocrinol Metab. 2009;4(5):435-442. doi:10.1586/eem.09.27 2. Brandi ML, Clunie GPR, Houillier P, et al. Challenges in the management of tumor-induced osteomalacia (TIO). Bone. 2021;152:116064. doi:10.1016/j.bone.2021.116064 3. Dahir K, Zanchetta MB, Stanciu I, et al. Diagnosis and management of tumor-induced osteomalacia: perspectives from clinical experience. J Endocr Soc. 2021;5(9):bvab099. doi:10.1210/jendso/bvab099 4. Minisola S, Peacock M, Fukumoto S, et al. Tumour-induced osteomalacia. Nat Rev Dis Primers. 2017;3:17044. doi:10.1038/nrdp.2017.44 5. Aono Y, Yamazaki Y, Yasutake J, et al. Therapeutic effects of anti-FGF23 antibodies in hypophosphatemic rickets/osteomalacia. J Bone Miner Res. 2009;24(11):1879-1888. doi:10.1359/jbmr.090509